| |
H2O2 is a nonplanar molecule with the bond lengths and bond angles as shown in fig 9.8. |
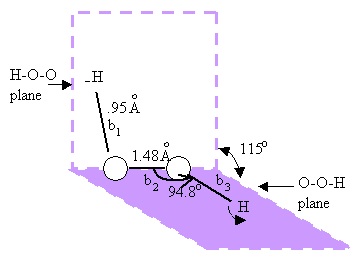 |
| Figure 9.8 The structure of H2O2. |
The angle between the first HOO plane and the next connecting OOH plane is 115o and this is called the dihedral angle. The different bond lengths in H2O2.are shown in fig 9.8. Methane is tetrahedral, XeF4 is planar (with the two lone pairs on either side of the XeF4 molecular plane), PCl5-, trigonal bipyramidal, SF4, a distorted terahedron and ClF3, T shaped. In the latter two cases, the lone pair - bond pair repulsions cause the distortion from regular shapes. The hybridization in both PCl5 and ClF3 may be thought of as sp3d or sp2pd with the three electron pairs of the sp2 group lying in a plane and the two electron pairs of the pd group lying perpendicular to this plane. One should remember not to get carried away by electron pairing or close shell concepts. |
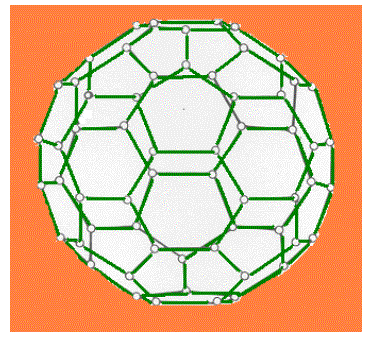
| During bonding, the shells of atoms can easily expand to include higher available orbitals. For example, in IF7 the structure is pentagonal bipyramidal with five atoms lying in a plane and the other two at the apices of the bipyramid. C60, buckminsterfullerene has the shape of a truncated icosahedron (appears like a football, of course, it is a molecular football ! ) with 20 hexagonal faces and 12 pentagonal faces. For a molecule with 60 atoms, it is one of the most symmetrical. |
|
|
|
| Fig 9.9 (a) The structure of C60 |
| |
|
| |
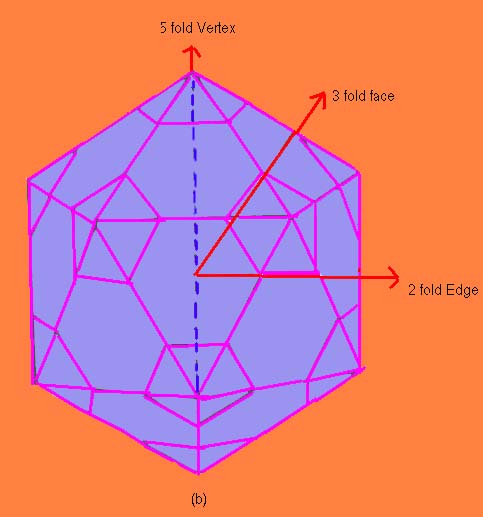
| Figure 9.9 (b) Symmetry elements of C60 |
| |
|
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
|