We have already studied the overlap diagrams of s and p orbitals in lectures 7 and 8. We shall consider the overlap diagrams of d orbitals here. Just as s and p orbitals combine to form
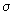 , , 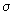 * , * ,
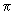 and and 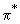 orbitals, d orbitals can also overlap to give bonding and antibonding combinations. We will consider combinations between two d orbitals of two atoms. Combinations between d and s and d and p can be constructed by analogy. orbitals, d orbitals can also overlap to give bonding and antibonding combinations. We will consider combinations between two d orbitals of two atoms. Combinations between d and s and d and p can be constructed by analogy.
|