The resonance structures do not imply that the molecule is present as one of these at various times. The actual structure is a combination of all these structures (with the first two contributing predominantly). In benzene, all the adjacent C-C bond lengths are identical and none of the structures in fig. 9.12 satisfy this criterion individually. |
|
| How are the coefficient of MOs and their energy levels obtained? |
| |
| You must have been wondering how the coefficients like 0.37 and 0.60 in butadiene or the energy levels in benzene can be determined. Each one of the MOs can be represented as |
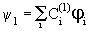 |
| |
| 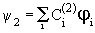 , ,
|
where the subscript in
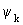 refers to the kth MO and the superscript i in the Ci(k) again refers to the kth MO, i.e., it is the coefficient of the ith atomic orbital in the kth MO. Each one of the MOs satisfies the Schrödinger equation. refers to the kth MO and the superscript i in the Ci(k) again refers to the kth MO, i.e., it is the coefficient of the ith atomic orbital in the kth MO. Each one of the MOs satisfies the Schrödinger equation. |
| |
H 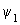 = = 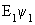 |
H 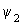 = = 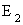 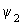 |
| These can be combined into the following determinantal equation. |
det | Hij - ESij | = 0, where Hij =
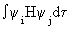 and Sij = and Sij =
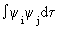 are elements of the matrices for H and S. The symbol det | M | refers to the determinant of the matrix M. are elements of the matrices for H and S. The symbol det | M | refers to the determinant of the matrix M. |
| |
| For butadiene, the above equation is a 4 x 4 equation which can be solved for the four energies E1, E2, E3 and E4. By substituting each energy in the above two equations, the values of the coefficients are obtained. The central theme in MO theory is to get good coefficients for the MOs, using improved forms of the above equations. |
| |