Problems |
(9.1) |
What is the angle between the two sp hybrid orbitals on a carbon atom of acetylene? What are the energy levels of these two hybrids? |
| |
|
| (9.2) |
Construct the antibonding combinations corresponding to figs 9.5 (a) , (b) and (c). |
|
|
| (9.3) |
Draw the overlap diagrams for bonding and antibonding combinations between (a) a p and an s orbital (b) between a d and an s orbital and (c) between a p and a d orbital. Indicate whether the MOs are 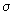 , , 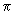 , etc. , etc. |
|
|
| (9.4) |
We discussed the resonance stabilization in benzene. Will there be a resonance stabilization in butadiene? Draw the resonance structures in butadiene. |
| |
|
| (9.5) |
Draw the resonance structures in SO3 . |
| |
| (9.6) |
NO3- and CO32- are electronic with (i.e., having the same number of electrons as in ) SO3 . What are the bond angles and bond orders in these molecules? |
| |
| (9.7) |
Calculate the value of the 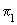 MO in benzene at the center of the molecule and at MO in benzene at the center of the molecule and at
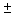 1 a0 on the molecular axis above and below the molecular plane. Assume each C-C bond length to be 1.35 1 a0 on the molecular axis above and below the molecular plane. Assume each C-C bond length to be 1.35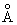 . . |
| |
| |
|
| (9.8) |
We have considered the bonding in linear H3 in an earlier lecture. Draw the MO overlap diagram for the lowest MO in triangular H3 |
| |
|
| (9.9) |
What is the expected hybridization on the phosphorous atom in PCl5 and PCl3? |
| |
|
| (9.10) |
What are the values of energies of a) sp, b) sp2 and c) sp3 hybrid orbitals in terms of the energies of s and p orbitals? Always remember that each hybrid orbital has to be normalised. E.g., 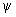 1 = 1 = 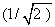 = (s + px) for a sp hybrid, because = (s + px) for a sp hybrid, because 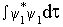 = 1 = 1 |
| |
|
| (9.11) |
Is an electron the same as its orbital? What is the difference between an orbital of a molecule and its wavefunction? |
| |
|
| (9.12) |
What is the electronic configuration of Mg? What is the hybridization in Mg in MgCl2? |
| |
|
| (9.13) |
Does water use sp2 hybrid orbitals or sp3 hybrids? justify your answer. |
| |
|
| (9.14) |
What is the FBF bond angle in BF3 and the HNH bond angle in NH3? What are the hybridizations in B and N in these two molecules? |
| |
|
| (9.15) |
If the three double bonds in benzene did not interact at all and contributed separately to the binding energy of benzene, then the contribution to the binding energy of benzene would be thrice the value of the energy of the C = C double bond in ethylene. From the MO energy level diagram of benzene we see that the energies of the three electron pairs are not same and the lowest MO gives a lot more stabilization than the double bond in ethylene. This additional stabilization in molecules is called the resonance energy. In which of the following molecules do you expect to have resonance energy (or resonance stabilization) ? Justify your answer |
| |
a) butadiene, b) hexatriene, c) 1,5 hexadiene, d) NO2, e) CO2 f) buckminsterfullerene (C60 ) and g) cyclopropene |
| |