It is instructive to study the bonding in unsaturated compounds as it gives good insights into the nature of MOs. It is convenient to study the 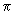 bonding independent of the sigma bonds which can be treated as constituting the "rigid" framework of the molecule. In ethene, for example, the bonding independent of the sigma bonds which can be treated as constituting the "rigid" framework of the molecule. In ethene, for example, the 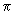 bond is in the plane perpendicular to the molecular plane. In butadiene, all the carbons and hydrogens lie in a plane and all the bonds in the plane are bond is in the plane perpendicular to the molecular plane. In butadiene, all the carbons and hydrogens lie in a plane and all the bonds in the plane are 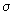 bonds. The bonds. The 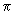 bonds are in a plane perpendicular to this plane and are formed from the four p orbitals (one on each of the four carbon atoms) perpendicular to the plane. The four bonds are in a plane perpendicular to this plane and are formed from the four p orbitals (one on each of the four carbon atoms) perpendicular to the plane. The four 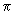 bonds that can be constructed from the four p orbitals (represented as bonds that can be constructed from the four p orbitals (represented as
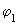 , ,
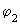 , ,
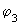 and and
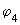 in the following equation) are shown in the following figure (fig 9.10). in the following equation) are shown in the following figure (fig 9.10).
|