The calculation of the contours for 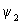 is similar to that of is similar to that of 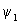 except that there are different signs of contours in different regions. The plots of except that there are different signs of contours in different regions. The plots of 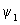 and and 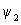 involved s orbitals on atoms A and B. We can now use the linear combinations using p orbitals. The four combinations that can be made are involved s orbitals on atoms A and B. We can now use the linear combinations using p orbitals. The four combinations that can be made are |