Here
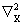 is the kinetic energy operator for nucleus X, is the kinetic energy operator for nucleus X,
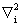 is the kinetic energy operator for electron i, riX is the distance between electron i and nucleus X and rij is the distance between electron i and electron j. Since the nuclei are held fixed, the nuclear kinetic energy operator terms will be set to zero. is the kinetic energy operator for electron i, riX is the distance between electron i and nucleus X and rij is the distance between electron i and electron j. Since the nuclei are held fixed, the nuclear kinetic energy operator terms will be set to zero. |