We have been considering all along, the behaviour of the MO, [eq (7.4)] which is called a bonding orbital. It is refered to as bonding because it increases the charge density between the nuclei (relative to separately incremented charge densities of 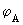 and and 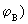 . Another MO, which does the opposite of this MO is called the antibonding MO as it decreases the electron density between the nuclei (in comparison to the non interacting or independently contributing AOs) and it is defined as . Another MO, which does the opposite of this MO is called the antibonding MO as it decreases the electron density between the nuclei (in comparison to the non interacting or independently contributing AOs) and it is defined as
|