As expected the energy of the bonding MO lies below the energy of the antibonding MO. Lower or more negative value of energy implies greater binding or greater stability. It is seen from figure 7.9 that the aufbau principle for these diatomics may be stated as 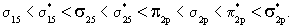 The ordering in the energies of The ordering in the energies of 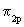 and and 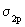 undergoes a change in O2 and F2 (compared to Li2 , Be2 ,
..up to N2) and here we take it as a fact. The reason for the change over is that in O2 and F2 the difference of the energies between undergoes a change in O2 and F2 (compared to Li2 , Be2 ,
..up to N2) and here we take it as a fact. The reason for the change over is that in O2 and F2 the difference of the energies between 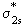 and and 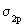 is very large so as not to affect them. If these energy levels are very near, these orbitals(which are of the same symmetry) would repel each other, pushing is very large so as not to affect them. If these energy levels are very near, these orbitals(which are of the same symmetry) would repel each other, pushing 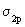 to go higher than to go higher than 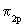 as in the case of Li2 , Be2 ,
..up to N2 . as in the case of Li2 , Be2 ,
..up to N2 .
|