| The Boltzmann Distribution of energies |
| |
The dependence of the probability of the earlier section on molecular speed can be re-expressed alternatively in terms of kinetic energy K.E = 1/ 2 m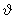 2. The probability 2. The probability 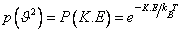 . The gas molecules in an ideal gas possess only kinetic energies and the dependence of the probability on . The gas molecules in an ideal gas possess only kinetic energies and the dependence of the probability on 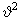 and on K.E are equivalent. However, in a general case, molecules possess both kinetic and potential energies (PEs) and the total energy E = K.E + P.E. Boltzmann generalised Maxwell's result for only KEs to include all energies and the probability distribution for energies, P(E), is called the Boltzmann distribution and is given by, and on K.E are equivalent. However, in a general case, molecules possess both kinetic and potential energies (PEs) and the total energy E = K.E + P.E. Boltzmann generalised Maxwell's result for only KEs to include all energies and the probability distribution for energies, P(E), is called the Boltzmann distribution and is given by, |
|
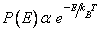 |
(29.31) |
This also has to be normalized such that the sum of P(E)s for all energies should be 1. Boltzmann, and Gibbs in particular, developed very general methods for deriving equations such as eq. (29.31) and here, we have merely stated the equation and given some justification for it. |
| As the energies of all microscopic systems are quantized into levels Ei, we should indeed be looking for the probability of finding a given system with energy Ei and this is given by |
|
|
 |
(29.32) |
The denomination in the above equation is a factor that ensures that
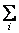 P(Ei) = 1 and it is called the partition function Q. P(Ei) = 1 and it is called the partition function Q. |
| |
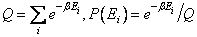 |
(29.33) |
Here,
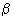 = 1/ k B T and is called the Boltzmann factor. = 1/ k B T and is called the Boltzmann factor. |
| Once we know the probability distribution for energy, we can calculate thermodynamic properties like the energy, entropy, free energies and heat capacities, which are all average quantities. To calculate P(E)s we need the energy levels of a system. The energy "levels" of a system can be built up from the molecular energy levels and intermolecular forces. We will consider the simpler problem of molecular energy levels which are pictorially shown in the figure 29.3. |
| |