Just as U is measured relative to U(0), A is measured relative to A(0). As T 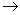 0, U(0) = A(0). 0, U(0) = A(0).
To obtain the thermodynamic functions, enthalpy (H) and the Gibbs free energy (G), we need to express pressure in terms of the partition function. From thermodynamics, we know that 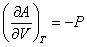 . Using this in eq (29.58), we get . Using this in eq (29.58), we get |