We have already commented that molecules have to collide with sufficient speeds so that the reaction occurs. Where does this sufficient energy come from? At a given temperature, not all molecules have the same speeds. During intermolecular collisions, these speeds change too. If we represent a molecular velocity by
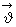 , it has three independent components , it has three independent components
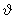 x, x,
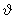 y and y and
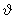 z in the three directions x, y and z. Let us consider monoatomic gas of molecular mass m. The probability F ( z in the three directions x, y and z. Let us consider monoatomic gas of molecular mass m. The probability F ( 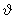 x, x, 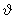 y, y, 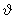 z) that a given molecule will have velocity components lying between z) that a given molecule will have velocity components lying between 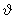 x and x and 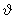 x+ d x+ d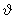 x, x, 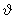 y and y and 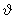 y + d y + d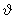 y and y and 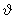 z and z and 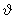 z + d z + d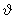 z is written as z is written as
|