You have already studied molecular energy levels in the spectroscopy section (Module 3). The electronic energy levels are widely separated. In each electronic level, there are several vibrational levels and for each vibrational level, there are several rotational states. This is a simplified and useful model to start with. The total energy is a sum of all these energies and is given by |
|
| E total = E electronic + E vibrational + E rotational + E translational + E others |
(29.34) |
The term Eothers includes nuclear spin energy levels and interactions between the first four. Assuming the first four to be independent and neglecting the last term, the molecular partition function (i.e., a sum over the molecular energy states) is given by |
| |
| q =
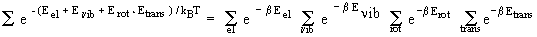
|
(29.34) |
| |
or q = q el q
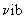 q rot q trans , where q rot q trans , where 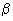 = = 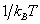 |
(29.35) |
| The molecular partition q function is written as the product of electronic, vibrational, rotational and translational partition functions. |
| |
The partition function is a sum over states (of course with the Boltzmann factor
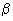 multiplying the energy in the exponent) and is a number. Larger the value of q, larger the number of states which are available for the molecular system to occupy. Since Eel > E multiplying the energy in the exponent) and is a number. Larger the value of q, larger the number of states which are available for the molecular system to occupy. Since Eel > E 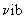 > Erot > Etrans, there are far too many translational states available compared to the rotational, vibrational and electronic states. qel is very nearly unity, q > Erot > Etrans, there are far too many translational states available compared to the rotational, vibrational and electronic states. qel is very nearly unity, q 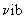 and qrot are in the range of 1 to 100 while qtrans can be much in excess of 1010. We shall calculate the values of these qs and indicate how these qs and the molecular velocity distribution functions are useful in calculating the rate constants. and qrot are in the range of 1 to 100 while qtrans can be much in excess of 1010. We shall calculate the values of these qs and indicate how these qs and the molecular velocity distribution functions are useful in calculating the rate constants. |
| |