In the present lecture, the temperature dependence of reaction rates was the focus of interest. Arrhenius equation accounted for the increase in reaction rates with increasing temperature with considerable success. It postulated an energy of activation which was the minimum energy needed for the reactants to cross over to the products. Temperature dependence of reaction rates was used to estimate the activation energy using the Arrhenius plot. The reaction coordinate is a parameter or an indicator indicating the extent of passage from the reactant to the product. Since the activation energy plays a critical role in reaction rates (except in diffusion limited reactions wherein just bringing the reactants “close to one another completes the reaction) we need to find out where this “extra” energy comes from. Additional energy may either come from collisions with molecules with great speeds or from some of the molecules which have excess internal energy which can be transferred to the reacting molecules. This led us naturally to the velocity distributions and energy distributions among molecules. We presented approximate derivations of the Maxwell-Boltzmann distribution of molecular speeds and the Boltzmann distribution of energies. The energy distributions are expressed in terms of partition functions which measure the number of available states with each state weighted by the Boltzmann factor, exp 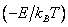 . Since the total molecular energy can be written as a sum over electronic vibrational, rotational and translation energies, the total partition function was expressed as the product of electronic, rotational, vibrational and translational partition functions. Separate expressions were obtained for these partition functions their estimates were given and the partition function for a collection of molecules and the average energy of a system was obtained in terms of partition functions. . Since the total molecular energy can be written as a sum over electronic vibrational, rotational and translation energies, the total partition function was expressed as the product of electronic, rotational, vibrational and translational partition functions. Separate expressions were obtained for these partition functions their estimates were given and the partition function for a collection of molecules and the average energy of a system was obtained in terms of partition functions. |