Problems |
29.1)
|
The rate constants for the decomposition of acetaldehyde are 0.343 M-1 s -1 and 0.789 M -1 s -1 at 70 K |
| |
and 81 K respectively. Find the preexponential factor and the activation energy. |
| |
29.2) |
Using the following information, find Ea and A ( in M -1 s -1 where M = mol / lit) |
| |
T in K |
300 |
350 |
400 |
450 |
|
|
|
|
|
k /(10 7 M 1 s 1) |
0.79 |
3.0 |
8.0 |
17 |
|
| |
|
| |
At what temperature is the rate constant at 300 K doubled? |
| |
|
| 29.3) |
The rate constant for a first order reaction is 4.53 * 10 8 and 5.93 * 10 9 s -1 at 363 K and 383 K |
| |
respectively. Find Ea and A (now in s -1). |
| |
|
| 29.4)
|
Bromination of propanone in acidic medium gave the following data. Find A and Ea. |
| |
|
| |
T / K |
293 |
297 |
300 |
302 |
k(10 6 M 1 s 1) |
1.16 |
2.02 |
5.59 |
8.60 |
|
| |
| 29.5) |
For the Maxwell - Boltzmann distribution of molecular speeds, at what speed is F(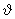 ) maximum? ) maximum? |
| |
| 29.6) |
At T = 300 K, for what 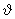 ( (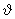 = max) is F ( = max) is F (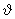 ) maximum? What should be the increase in T so that the ) maximum? What should be the increase in T so that the |
new F (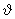 max) ( at the higher T) has half the value of F ( max) ( at the higher T) has half the value of F (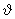 max) at 300 K? max) at 300 K? |
| |
| 29.7) |
The ground state of Na is a doublet (two states with the same energy). Assuming this to be the zero of |
| energy and assuming that the next higher energy to be 2 e V higher than this state, calculate q e. |
| |
| 29.8) |
The bond length e of O2 is 1.2
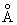 . The moment of inertia I is m r 2e / 2 where m of O is 16 * 1.66 * 10 -27 . The moment of inertia I is m r 2e / 2 where m of O is 16 * 1.66 * 10 -27 |
| |
kg. Calculate B and the rotational partition function at 300 K. |
| |
| 29.9) |
The vibrational frequency of ICl is 384 cm -1 . What is its vibrational partition function at 300 K ? What is |
| |
the fraction of molecules in the ground state (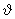 = 0) and the first excited state = 0) and the first excited state 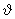 = 1? = 1? |
| |
| 29.10) |
Calculate the translational partition function of N2 at 300 K. For volume, use the molar volume at 300 K. |
| |