| |
This implies that the spectrum is independent of the value of K. The physical meaning is as follows. K refers to the rotation about the symmetry axis such as the C - Cl axis. A rotation about this axis does not change the dipole moment. We mentioned in the section on the rotational spectra of diatomics that the molecular dipole moment has to change during the rotational motion (transition dipole moment operator of Eq 12.5) to induce the transition. Rotation along the axis A and B changes the dipole moment and thus induces the transition.
By using rotational or microwave spectroscopy, very accurate values of bond lengths can be obtained. For example, in HCN, the C-H length is 0.106317 ± 0.000005 nm and the CN bond length is 0.115535 ± 0.000006 nm. The principle of the microwave oven involves heating the molecules of water through high speed rotations induced by microwaves. The glass container containing water however remains cold since it does not contain rotating dipoles. |
13.4 |
Vibrations and Rotations of a diatomic |
| |
You have noticed in your earlier studies that simple pendulums or stretched strings exhibit simple harmonic motion about their equilibrium positions. Molecules also exhibit oscillatory motions. A diatomic oscillates about its equilibrium geometry. The quantized vibration energies Eυ of a harmonic oscillator are
Eυ = (u + ½) hν (13.20)
v = 0,1,2,…………
The vibrational frequency ν is related to the force constant k through
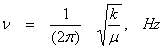 (13.21) (13.21)
The vibrational motion occurs under the action of a binding potential energy. The potential energy (PE) curve for a harmonic oscillator is given in Fig. 13.4. |
| |
Figure 13.4. The potential energy of a harmonic oscillator V = k(r-ro)2. The force constants k in N/m for a few moleculues are, CO (1902), HF (966), HCl (516), HI (314). |