| |
In CH3Cl for example, the main symmetry axis is the C – Cl axis. We need two quantum numbers to describe the rotational motion with respect to IA and IC respectively. Let J represent the total angular momentum of the molecule and K the angular momentum with respect to the C – Cl axis of the symmetric top. J takes on integer values and K can not be greater than J (recall that ml≤ l | for orbital angular momentum). The (2J + 1) “degeneracy” is expressed through the 2J + 1 values that K can take.
K = J, J – 1, ….. 0, - (J – 1), - J (13.15)
The rotational energies of a symmetric top are given by
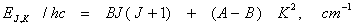 (13.16) (13.16)
The moments of intertia are related to B and A as
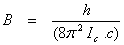 and and 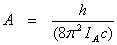 (13.17) (13.17)
As the energy depends on K2, energies for states with + K and – K are doubly degenerate. Thus there will be
J + 1 levels and (2J + 1) states for each values of J.
The selection rules for the symmetric top are,
ΔJ = ± 1 and ΔK = 0 (13.18)
It can be easily shown hat
|
| |
(EJ +1, K – E J,,K)/hc = 2 BJ (J +1) (13.19) |