| |
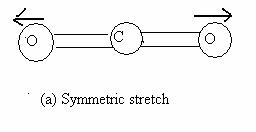
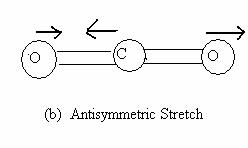
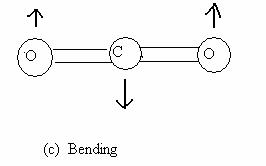
The three normal modes of vibrations of water (Fig 13.7) are the symmetric stretch (ν1 = 3651.7 cm-1), the antisymmetric stretch (ν2 = 3755.8 cm-1) and the symmetric bend (ν3 = 1595.0 cm-1). Bending requires less energy and thus, its frequency is lower. The asymmetric stretch requires greater reorganization than the symmetric stretch and hence a larger frequency. Molecular CO2 is a linear triatomic and has 3N - 5 = 4 normal modes of vibration. The symmetric stretch (ν1 = 1330 cm-1) asymmetric stretch (ν2 = 2349.3 cm-1) and bending (ν3 = 66.3 cm-1) are shown in Fig 13.8. The bending mode is doubly degenerate, owing to the two independent bending modes in two perpendicular planes containing the molecular axis.
|
| |
Figure 13.8 Normal modes of vibrations of CO2 |
| |
Different molecules can be easily identified by their normal mode frequencies. In addition to these modes, overtones (2ν1, 3ν2, etc.), combination bonds (ν1 + ν2, 2ν1 + ν2, ν1 +ν2 +ν3 …), and difference bands (ν1 - ν2, ν1 +ν2 - ν3) can be observed. Since a large number of rotational and vibrational levels are closely spaced they provide a rich base for setting up lasers when the upper levels are populated. As in the case of diatomics, rotational lines are richly dispersed in vibrational spectra of polyatomics. The concept of normal modes can be extended to solids and liquids too. Since in a solid, there are a very large number of atoms (of the order of Avogadro number), there are 3N-6 normal modes. These are characterized as phonons, which correspond to collective motions of atoms in a solid. |