| |
The quantized rotational energy levels for this diatomic are
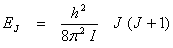 (13.7) (13.7)
The energy differences between two rotational levels is usually expressed in cm-1. The wave number corresponding to a given ∆E is given by
ν = ∆E /hc, cm-1 (13.8)
The energy levels in cm-1 are therefore,
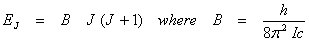 (13.9) (13.9)
The rotational energy levels of a diatomic molecule are shown in Fig. 13.2. |
| |
Fig. 13.2 Rotational energy levels of a rigid diatomic molecule and the allowed transitions. |
| |
The selection rule for a rotational transition is,
∆ J = ± 1 (13.10) |