| |
The center of mass is defined by equating the moments on both segments of the molecular axis. |
| |
The moment of inertia is defined by |
| |
Since 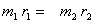 = = 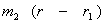 , , 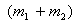 r1 = m2r Therefore, r1 = m2r Therefore,
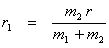 and and 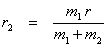 (13.4) (13.4)
Substituting the above equation in (13.3), we get
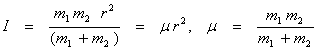 (13.5) (13.5)
Where μ, the reduced mass is given by
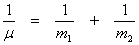 (13.6) (13.6)
The rotation of a diatomic is equivalent to a “rotation” of a mass μ at a distance of r from the origin C. The kinetic energy of this rotational motion is K.E. = L2/2I where L is the angular momentum, Iω where ω is the angular (rotational) velocity in radians/sec. The operator for L2 is the same as the operator L2 for the angular momentum of hydrogen atom and the solutions of the operator equations L2 Υlm = l (l + 1) Ylm, where Ylm are the spherical harmonics which have been studied in lecture 3. |