| |
Often, one observes a combined vibrational rotational spectrum. A combined set of vibrational and rotational energy levels of a diatomic is given by
Etotal = BJ (J + 1) + (v + ½ ) ν - xe (v+ ½)2 ν, cm-1 (13.25)
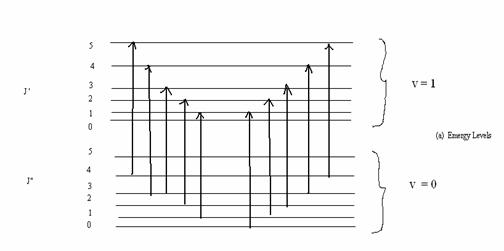
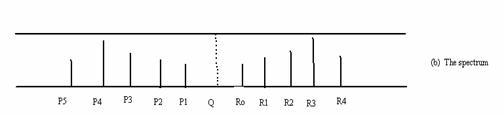
The energy level diagram and the spectrum corresponding to the diagram are shown in Fig. (13.6).
|
| |
Fig. 13.6. The vibrational rotational spectrum.
|
| |
The selection rules are ∆v = ± 1, ± 2,…
∆ J = ± 1. ∆ J = 1 corresponds to the R branch on the right at higher frequencies and
∆ J = J” – J’ = -1 corresponds to the P branch on the left. The dashed line Q for which
∆ J = 0, is not seen. The difference between R0 and P1 is 4B and the difference between adjacent R lines and adjacent P lines is 2B. |