Thus the apparent frequency is
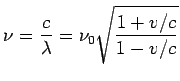
For non-relativistic velocities, the Doppler shifted frequencies are
(It may be noted that in this case there is a Doppler effect in the transverse direction as well. However, the effect being of the order 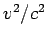 , is neglected.) Doppler shift may be significant in gases in which atoms move randomly due to thermal motion. According to the kinetic theory of gases, the distribution of velocities of atoms (or molecules) in a gas at temperature , is neglected.) Doppler shift may be significant in gases in which atoms move randomly due to thermal motion. According to the kinetic theory of gases, the distribution of velocities of atoms (or molecules) in a gas at temperature 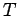 is given by Maxwellian velocity distribution is given by Maxwellian velocity distribution
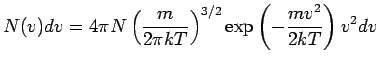
where 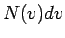 is the number of atoms with speeds between is the number of atoms with speeds between 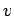 and and 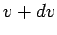 . All other symbols in the equation have their standard meaning as in the kinetic theory. The average speed of the molecules is given by . All other symbols in the equation have their standard meaning as in the kinetic theory. The average speed of the molecules is given by
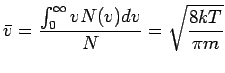 |