Example 6:
Starting with n0 moles of water which dissociates into H2
and O2, show that at equilibrium
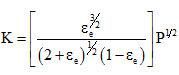
At an average temperature of 1900K, the slope of the graph ![]() Â
for dissociation of water vapour is found to be -13000. Find the heat of dissociation.
Is it an exothermic or endothermic reaction?
Â
for dissociation of water vapour is found to be -13000. Find the heat of dissociation.
Is it an exothermic or endothermic reaction?
Solution:
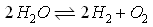

Now
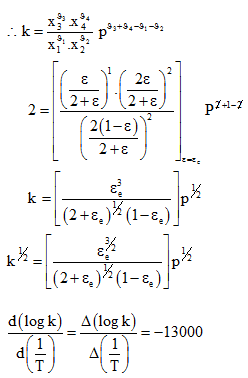
Again van’t half equation
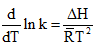
By rearrangement, we get,
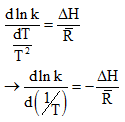
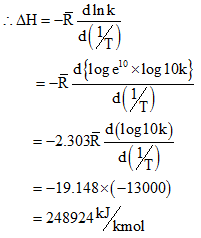
Since ΔH is positive, hence the reaction is endothermic.