Unsolved Problems
- Calculate the degree of ionization of cesium vapour at 10-6 atm at the two temperatures of 2260 and 2520 K.
- The volumetric composition of the ‘dry’ products of combustion of an unknown hydrogen fuel. CxHy , gives
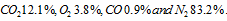 . Determine (a) the chemical formula of fuel, (b) the air fuel ratio, and (c) the percentage of excess air used.
. Determine (a) the chemical formula of fuel, (b) the air fuel ratio, and (c) the percentage of excess air used. - Liquid octane is burned with air in a combustion test, and the dry volumetric analysis of the products reveals the following composition:
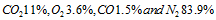 .compute the actual air-fuel ratio used during the test and the percentage of excess air used.
.compute the actual air-fuel ratio used during the test and the percentage of excess air used. - Carbon monoxide and 300% theoretical air enter a steady flow combustor at 400 K and a low pressure. The energy released by the reaction heats the product gases to 1400 K. If the combustion is complete, estimate the heat gained or lost through the walls of combustor.
- A mixture of methane and oxygen, in the proper ratio for complete combustion and at 25°C and 1 atm, reacts in a constant volume calorimeter bomb. Heat is transferred until the products of combustion are at 400 K. Determine the heat transfer per mole of methane.
- Gaseous butane at 25°C is mixed with air at 400 K and burned with 400% theoretical air, determine the adiabatic flame temperature.
- Octane gas at 25°C, 1 atm enters a jet engine and burns completely with 300% theoretical air at 25°C, 1 at. Products of combustion leaves at 990K, 1 atm. If the fuel and air enter with relatively low air velocities, determine the velocity of the combustion products at exit. Neglect PE effects and heat transfer between the engine and surroundings.
- Liquid octane (C8H18) at 25°C, 1 atm enters an adiabatic reactor operated at steady and burns completely with air entering at 227°C, 1 atm. If the combustion products exit at 1127°C, determine the percent excess air used, neglecting KE and PE effects.