Example 9:
A mixture of η0 , ν1 moles of A1 η0,
ν2 moles of A2 at temp T and pressure p occupies a volume
V0, when the reaction is ;
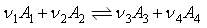
has come equilibrium at the same T and p, the volume is Ve , show
that
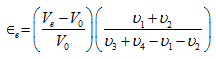
Solution:
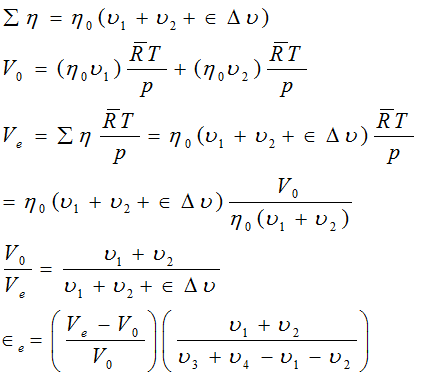
Example 10:
A gasoline engine delivers 150 kW. The fuel used is C8H18(l) and it enters the engine at 25°C. 150 % theoretical air is used and it enters at 45°C. The product of combustion leave the engine at 750 K, and the heat transfer from engines is 205 kW. Determine the fuel consumption per hour if complete combustion is achieved.
Solution:
The stoichiometric equation is given as;

Energy input from engine Wcv=150kW
Qcv=-205kW
Let n kg mol of fuel be consumed per second,
By first law,
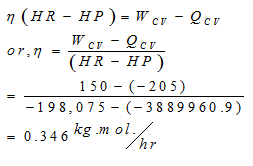
Therefore, Fuel consumption rate=η X molecular wt.
= 0.346 X 114
= 39.4 kg/hr