Example 4:
Calculate the equilibrium composition if argon gas is heated in an are to
10000 K, 1 kPa assuming the plasma to consist of A, A+, ![]() the equilibrium constant of the reaction at this temp is 0.00042
the equilibrium constant of the reaction at this temp is 0.00042
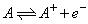
Solution
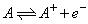

Now
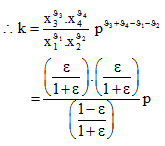
 ..............................................(1)
..............................................(1)
At T=10000K, p=1kpa
K=0.00042
Substituting the above in 1, we get
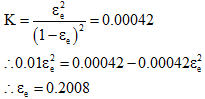
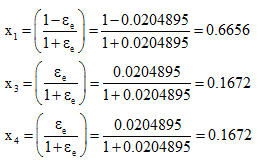
The composition is
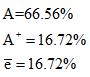
Example 5:
The combustion chamber of a gas turbine used a liquid hydrocarbon fuel which
has an approximate compostion of C8H18. On test the following data are obtained.
| Tair = 400 K | Tproduct = 1000 K |
| Vair = 100 m/s | Vproduct = 150 m/s |
| Tfuel = 50° C | FAactual = 0.0211 kgfuel/kgair |
Calculate the combustion efficiency.
Solution
For the ideal chemical reaction, the heat transfer is zero. Therefore, writing
the first law for a control volume that includes the combustion chamber, we
have,
![]()