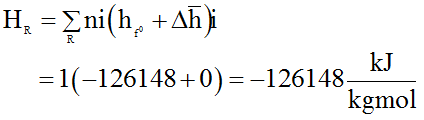Or, 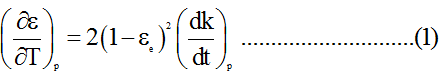
Now, for k=0.0174
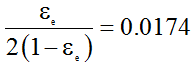
Or, 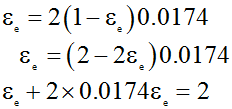
![]() .................................(2)
.................................(2)
Again from van’t hoff equation
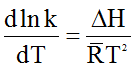
Or, 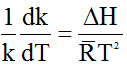
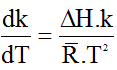 .............................................. (3)
.............................................. (3)
From 1, 2, 3 we get
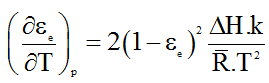
Substituting
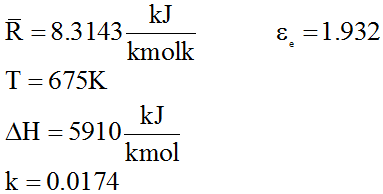
We get,
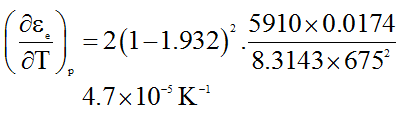
Example 2:
A mixture of butane and 150% theoretical air enters a combustion chamber at 250c, 100kpa and products of combustion leaves at 1000K, 100kpa. Determine heat transfer from the combustion chamber and irreversibility of the process?
Solution
The stoichiometric equation is
![]()
Actual combustion equation with 150% theoretical air is,
![]() Applying 1st law of thermodynamics,
Applying 1st law of thermodynamics,