3-5.7 .2.2 Indirect non-isotopic labeling involves chemical linkage of reporter molecule to a nucleotide. When this modified nucleotide is incorporated into DNA, then it is specifically bound to a protein or other ligand which has high affinity against the reporter group. Long spacer is introduced between nucleotide and reporter so as to reduce steric hindrances for binding of affinity molecule.
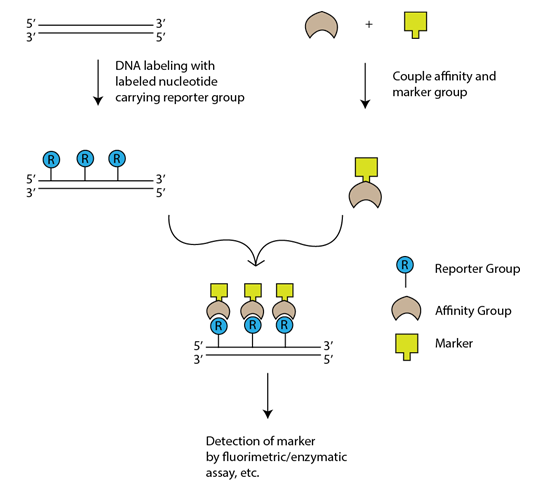
Fig. 3-5.7 .2.2: Indirect non-isotopic labeling
Two widely used non-isotopic labeling methods are:
Biotin-Streptavidin Method: This method uses two ligands which has high affinity towards each other: Biotin works as the reporter and the bacterial protein streptavidin is used as the affinity molecule. Biotinylated nucleotides like bio-11-dUTP are used as labeling agents with a spacer of 4-16 C atoms long between biotin and dNTP. However, Biotin is a ubiquitous constituent of mammalian tissues and tends to stick easily to certain type of nylon membranes which leads to high levels of background during in situ, northern and southern hybridization. To overcome this background problem, digoxigenin is used.
Digoxigenin, a plant steroid obtained from Digitalis plant and is used as a reporter and an affinity molecule. Digoxigenin is thus an all-purpose immuno-tag, and in particular a standard immunohistochemical marker for in situ hybridization.
Enzymatic methods are mostly used to label DNA probes with biotinylated nucleotides. Photochemical labeling of biotin to nucleic acids can also be used. The label is linked to nitrophenylazido group that is converted by UV irradiation to highly reactive nitrene that form stable covalent linkages to DNA or RNA.
End labeled DNA can be used as:
-
Molecular-weight standards in Southern blotting
-
Probes in gel-retardation experiments
-
Tracers for small quantities of DNAs on gels
-
Probes for screening bacterial colonies or plaques
-
Substrates for Maxam-Gilbert sequencing
-
Probes for RNA mapping with S1 Nuclease or Mung bean nuclease
-
Primers in primer-extension reactions.