| |
where 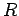 is called the Rydberg constant, which has a numerical value is called the Rydberg constant, which has a numerical value 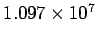 m m 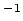 . . |
| |
The de Broglie hypothesis may be used to derive Bohr's formula by considering the electron to be a wave spread over the entire orbit, rather than as a particle which at any instant is located at a point in its orbit. The satble orbits in an atom are those which are standing waves. Formation of standing waves require that the circumference of the orbit is equal in length to an intergral multiple of the wavelegth. Thus, if 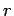 is the radius of the orbit is the radius of the orbit |
| |
which gives the angular momentum quantization |