| Objectives |
| In
this course you will learn the following |
 |
Matter at very small length scale behave like waves. |
 |
de Broglie hypothesis associates a wavelength 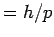 with matter waves. with matter waves. |
 |
Electron diffraction from crystals which confirm wave nature of electrons. |
 |
Bohr's model of hydrogen atom and its relationship with matter waves. |
 |
Double slit experiments performed with electrons give results similar to Young's experiment for light. |
 |
Heisenberg uncertainty principle. |