| |
Heisenberg Uncertainty Principle |
 |
The clue to this apparent paradox is in Heisenberg's uncertainty principle, according to which, there are limitations on the accuracy with which experiments aimed at determining which hole a particular electron goes through, may be performed, while still not disturbing the interference effect.
Classically, one can simultaneously measure the position and momentum of a particle to infinite precision. In the quantum mechanical world, there is an uncertainty associated with every measurement. Whenever we attempt to measure the position of an object with a precision 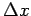 , there is some uncertainty , there is some uncertainty 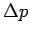 in the momentum measurement, which we cannot get rid of, because, in order to make a measurement we must disturb the system. in the momentum measurement, which we cannot get rid of, because, in order to make a measurement we must disturb the system. |
| |
In the thought experiment gedanken shown in the figure, the electron is assumed to be at restly below the objective of the microscope. The electron is illuminated by gamma rays, which have a short wavelength. Using a short wavelength yields high resolution. According to principles of optics, a microscope can resolve objects to within 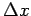 , which is related to the wavelength , which is related to the wavelength 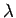 through through |