| |
Wave Nature of Particle - the de Broglie Hypothesis |
| |
In experiments like photoelectric effect and Compton effect, radiation behaves like particles. de Broglie, a french physicist asked whether in some situations, the reverse could be true, i.e., would objects which are generally regarded as particles (e.g. electrons) behave like waves ? In 1924 de Broglie postulated that we can associate a wave with every material object. In analogy with photons, he proposed that the wavelength 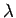 associated with such a matter wave is related to the particle momentum associated with such a matter wave is related to the particle momentum 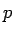 through the relationship through the relationship |
| |
where 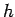 is the Planck's constant is the Planck's constant |