| |
The reason behind this paradox is that when we observed the electron, the electron has to interact with whatever probe we use for observing, in this case, with the light source. This interaction can alter the chance of arrival of the electron at the point P. |
| |
Can we use a weaker source of light to reduce the effect ? The answer clearly is no because a weaker source does not mean photon energy is different, it simply means that there are less number of photons. If there are less number of photons, some of the electrons will escape without being detected by them. Those which are detected are distributed according to |
(1) |
while those which escape being detected are distributed according to |
(2) |
and the net result that we get is a weighted mean of the two distributions. |
 |
Principle of Complementarity :
The above behaviour illustates an important principle of physics called the principle of complementarity. When we tried to determine which slit the electron went through, we were investigating the particle nature of an electron. However, only a wave can simultaneously go through both the slits and the interference effect was a manifestation of the wave nature. According to the principle of complementarity, an object has both the particle property and wave property. However, in a given experiment either one or the other property can be determined. It is not possible to siumultaneously get information on both the particle nature and the wave nature of an object in the same experiment. |
| |
What the photons do to the electron is to transfer momentum so that the electrons are scattered in different directions from their original directions. Can we then reduce the effect by using light of longer wavelength which can impart less momentum causing less disturbance ? The answer once again is no because if the wavelength is too long we will not be in a position to say whether the scattered electron came from the slit 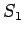 or from or from 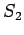 as the position cannot be determined with a precision better than the wavelength. as the position cannot be determined with a precision better than the wavelength. |