UNSOLVED QUESTIONS
- Two kg of water at 80°C are mixed with 3 (three) kg of water at 30°C in a constant pressure of 1 atmosphere. Find the increase in the entropy of the total mass of water due to mixing process (Cp for water = 4.187 kJ/kg-K)
- Two identical bodies of constant heat capacity are at the initial temperature Ti. A refrigerator operates between these two bodies until one body is cooled to temperature T2. If the bodies remain at constant pressure and undergo no change of phase, show that the minimum amount of work needed to do this is
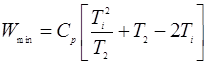
- A reversible heat engine shown in Fig. 1, during a cycle of operation draws 5 MJ from 400 K reservoir and does 840 KJ of work. Find the amount and direction of heat interaction with other reservoirs.
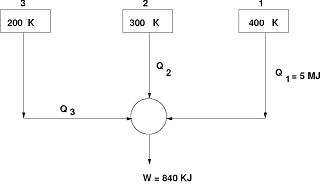
Fig. 1: Figure for problem 3
- Show that if two bodies of thermal capacity c1 and c2 at temperatures T1 and T2 are brought to the same temperature T by means of a reversible heat engine, then
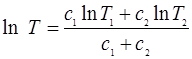
- An inventor claims to have developed an engine that takes in 107 J at a temperature of 400 K, rejects 4 X 105 J at a temperature of 200 K and delivers 3.6 X 105 Jof mechanical work. Would you advice investing money to put the engine on the market?
- A mass m of a liquid at a temperature T1 is mixed with an equal mass of the same liquid at temperature T2. The system is thermally insulated. Show that the change of the universe is
Also prove that this is necessarily positive.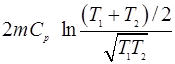
- A body of constant heat capacity Cp and at a temperature T1 is put in contact with a reservoir at a higher temperature Tƒ. The pressure remains constant while the body comes to equilibrium with the reservoir. Show that the entropy change of the universe is equal to
Prove that this entropy change is positive.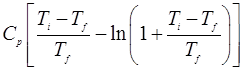
- An adiabatic vessel contains 2 kg of water at 25°C. By paddle wheel work transfer, the temperature of water is increased to 30°C. If the specific heat of water is assumed constant at 4.187 kJ/kg-K, find the entropy change of the universe.
- A resistor of 30 ohms is maintained at a constant temperature of 27°C while a current of 10 amperes is allowed to flow for 1 second. Determine the entropy change of the resistor and the universe.
- If the resistor ( See Problem 9) initially at 27°C is now insulated and the same current is passed for same time, determine the entropy change of the resistor and the universe. The specific heat of resistor is 0.9 kJ/kg-K and mass of resistor is 10 gm.
- The value of Cp of a certain substance can be represented by Cp = a + bT. Determine the heat absorbed and increase in entropy of a mass m of the substance when its temperature is increased at constant pressure from T1 to T2.
- The heat transfer rate from a very long fin of constant cross section is given by
where h is the convective heat transfer coefficient, P is the perimeter of the fin in a plane normal to its axis, kt is the thermal conductivity of the fin, and A is the cross-sectional area of the fin (again in a plane normal to its axis). T∞ is the temperature of the fin’s surrounding (measured far away from the fin itself) and Tf is the temperature of the base of the fin.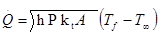
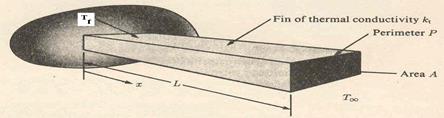
Fig. 2: A long fin ( Problem 12)
The temperature profile along the fin is given by
T(x) = T∞ + (Tf - T∞)e-mx
The fin is attached to an engine whose surface temperature is 95° C. determine the entropy generation rate for the fin if it is a very long square aluminium fin 0.01 m on a side in air at 20° C. The thermal conductivity of aluminium is 204 W/m.K and the convective heat transfer coefficient of the fin is 3.5 W/m2. K.
where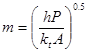
- At a certain location the temperature of the water supply is 288 K. Ice is to be made from this water supply by the process shown in Fig. 3. The final temperature of the ice is 263 K, and the final temperature of the water that is used as cooling water in the condenser is 303 K. What is the minimum work required to produce 1000 kg of ice?
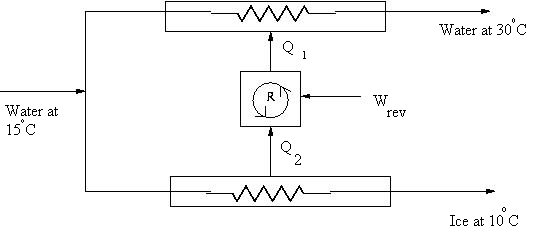
Fig. 3 - The velocity profile in the steady laminar flow of an incompressible Newtonian fluid contained between concentric cylinders in which the inner cylinder is rotating and the outer cylinder is stationary is given by
where ω is the angular velocity of the inner cylinder and x is measured radially outward. Determine the rate of entropy production due to laminar viscous losses for engine oil at 20° C in the gap between cylinders of radii 0.050 and 0.051 m when the inner cylinder is rotating at 1000 rev/min. The viscosity of the oil is 0.8 N.s/m2 and the length of the cylinder is 0.100 m.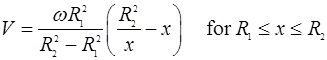
- A single tube, single pass heat exchanger is used to cool a compressed air flow of 0.2 kg/s from 90 to 75° C. The cooling fluid is liquid water that enters the heat exchanger at 20° C and leaves at 40° C. If the overall heat transfer coefficient is 140 W/m2K and all flow streams have negligible pressure drop, determine the required heat exchanger area and entropy production rate for
- Parallel flow, and,
- Counter flow.