NERNST-SIMON STATEMENT:
| The entropy change associated with any isothermal reversible process of a condensed system approaches zero.
|
Another statement of third law can be given like this:
It is impossible by any procedure, no matter how idealized, to reduce the entropy of a system to zero point value in a finite number of operations. |
Physical and chemical facts which substantiate the third law:
- For any phase change that takes place at low temperature, Clausius-Clayperon equation hold good,
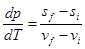
1.276
From third law of thermodynamics,
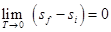 , since vf - vi is not zero. It shows that
, since vf - vi is not zero. It shows that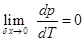
1.277
This is substantiated by all known sublimation curves.

Fig. 1.41 The temperature dependence of the change in the Gibbs function and in the enthalpy for an isobaric process ΔG = ΔH - TΔs
1.278
At low temperature, TΔS is very small.
∴ ΔG = ΔH
1.279
Which confirms that
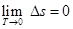 .
.- From the Gibbs-Helmholtz equation
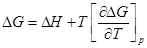
1.280
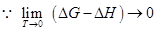 (Fig. 1.41)
(Fig. 1.41)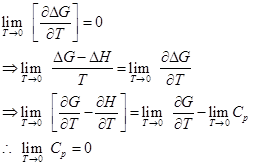
1.281
Similarly, using Gibbs Helmholtz equation containing U and F
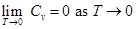
1.282
∴ Cp = Cv = 0 as T → 0
1.283