Marching towards absolute zero temperature.
Motivation
Application of first law and second law of thermodynamics to reactive systems become difficult due to the non availability of a standard reference entropy value of various substances. There is a need to have a reference entropy value for all substances for evaluating the efficiency of a reactive system. Third law of thermodynamics provides a base value for the entropy.
The third law of thermodynamics was formulated during the early part of twentieth century. The initial work was done primarily by W. H. Nernst [1864-1941] and Max Planck [1858-1947].
Attaining low temperature
- Below 5 K is possible by Joule Kelvin expansion, by producing liquid helium.
- Still lower temperature can be attained by adiabatic demagnetization of a paramagnetic salt.
- Temperature as low as 0.001 K has been achieved by magnetic cooling.
Magnetic Property
- Diamagnetic: substance is repelled by magnet
- Paramagnetic: substance attracted by magnet, such as Iron, Gadolinium sulphate
Adiabatic Demagnetization of a paramagnetic salt:
- Gadolinium sulphate is used here.
- Salt is hung by a fine nylon thread inside the salt tube such that it does not touch the sides.
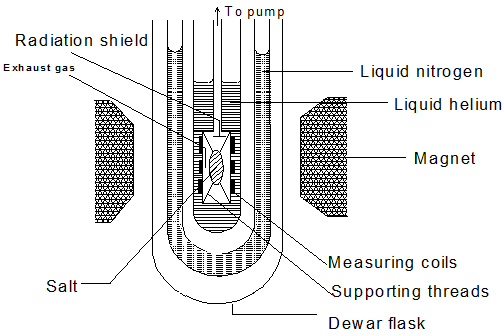
Fig: 1.39 Adiabatic demagnetization of a paramagnetic salt
- The salt is first cooled slightly below 1 K by surrounding it with liquid helium boiling under reduced pressure.
- Next, the salt is exposed to a strong magnetic field of about 25000 Gauss. Heat produced due to magnetization of the salt is transferred to the liquid helium without causing an increase in salt temperature.
- With the magnetic field still present, the inner chamber containing the salt is evacuated of gaseous helium.
- Finally, the magnetic field is removed. The molecules disalign themselves, which require energy. This energy is obtained by the salt getting cooler in the process.
- Repetition of the process lowers down the temperature of the salt. Temperature as low as 0.001 K have been achieved so far.
Measurement of temperature
In the neighborhood of absolute zero, all ordinary methods of temperature measurement fail. Curie’s law gives the most convenient method for measurement of at low temperature (approximately).
|
1.275 |
where ![]() is the magnetic susceptibility of the salt
is the magnetic susceptibility of the salt
T is the absolute temperature
c is the Curie’s constant.
The fundamental features of all cooling process are that the lower the temperature achieved, the harder it is to go still lower.