Interpretations of adiabatic demagnetization:
- Final temperature Tf ∞ Ti (initial temperature)
- First demagnetization produces a temperature half that at start
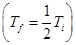
- Second demagnetization produces a temperature
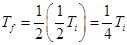
- Third demagnetization produces a temperature
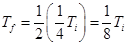
- rth demagnetization produces a temperature
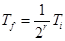
Infinite number of adiabatic demagnetizations will be required to attain absolute zero temperature.
FOWLER-GUGGENHEIM STATEMENT OF THIRD LAW:
It is impossible by any procedure, no matter how idealized, to reduce any condensed system to the absolute zero of temperature in a finite number of operations. |
Any isothermal magnetization from 0 to H (magnetic intensity) such as k - i1, f1 - i2 etc. is associated with the release of heat, i.e., a decrease in entropy (Fig. 1.40).
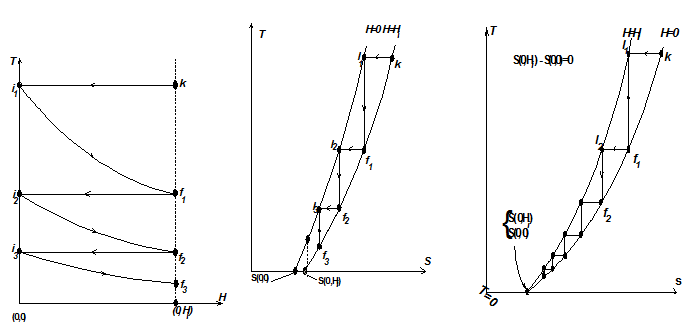
Fig. 1.40 T-H and T-S diagrams of a paramagnetic substance to show the equivalence of three statements of the third law
The processes i1 - f1, i2 - f2, i3 - f3 etc. represent reversible adiabatic demagnetizations with temperatures getting lower and lower. Repeated cycles of isothermal magnetization and adiabatic demagnetization would bring about a very low temperature. It is seen that [S(T, H) - S(T,0)] decreases as the temperature decreases, i.e., ΔSIII < ΔSII < ΔSI. Thus entropy change associated with an isothermal reversible process of a condensed system approaches zero. It is called Nernst-Simon statement of third law of thermodynamics.