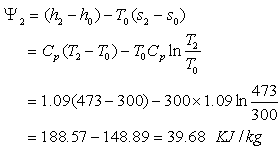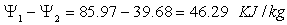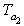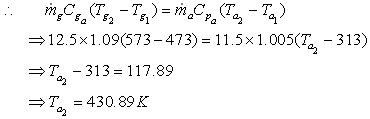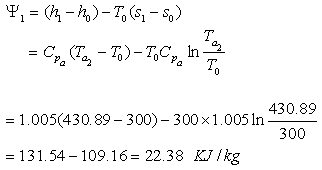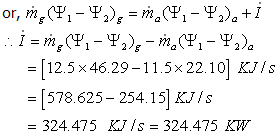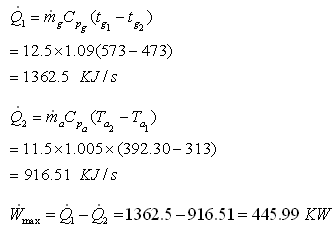Example 6: Water at 90°C flowing at the rate of 2 kg/s mixes adiabatically with another stream at 30°C flowing at the rate of 1 kg/s. Estimate the rate of entropy generation and rate of energy loss due to mixing. Take T0 = 300 K.
Solution:
Let Tf be the final temperature of the mixture. Since the mixing is occurring
adiabatically, no heat transfer takes place outside the system. However, heat
transfer takes place between the fluids only due to the temperature.
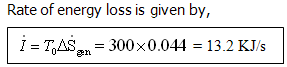
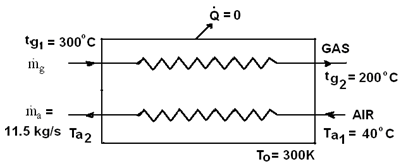
Example 7: An air pre-heater is used to cool the products of combustion from
a furnace for heating air to be used for combustion. The rate of flow of product
is 12.5 kg/s and product cool from 3000C to 2000C. Rate of air flow is 11.5
kg/s. Initial air temperature is 400C.
Find
(a) Initial and final availability of the products.
(b) What is the energy loss of the process?
(c) If the heat transfer from the products took place reversibly through heat
engines what would be the final temperature of the air?
(d) What power would be develop by the heat engine.
Take
T0 = 300K, Cpg = 1.09 KJ/kg-K, Cpg
=1.005 KJ/kg-K
neglecting pressure drop for fluid and heat transfer to the surroundings.
Solution:
Availability of gas at the point of entry to the heat exchanger
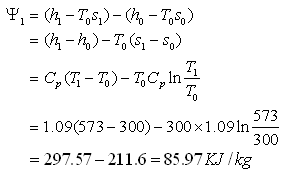
Availability of gas at exit,
Therefore decrease in availability of gas,
Similarly, for air
Let the final temperature of air be
Since there is no heat transfer outside the heat exchanger, heat given by
the gas is taken by the air.
Availability of air at the outlet,
Availability of gas at the inlet,
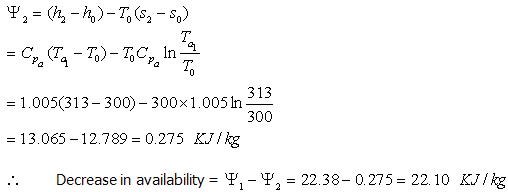
Energy loss of the gas = Energy gain of Air + Energy destruction
Part C
Heat is transformed from gas to air reversibly through large number of heat
engines.
Therefore entropy generation rate is zero.
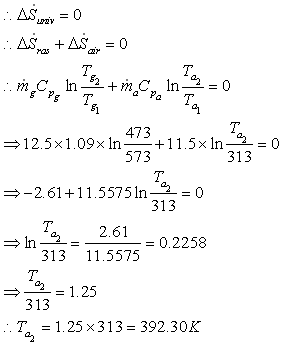
Part D