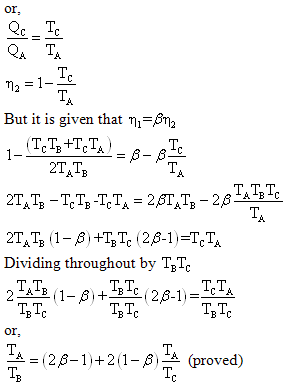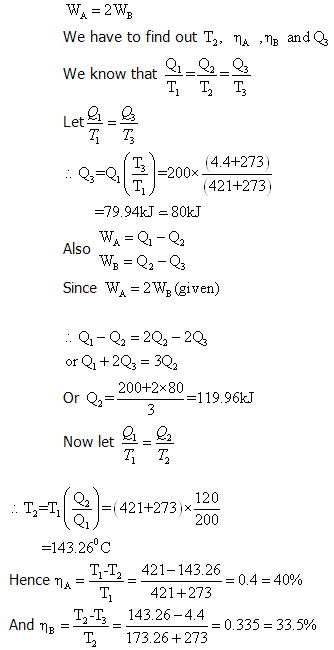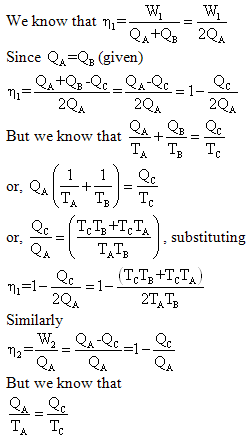Solved Problems
Example 1: Two reversible heat engines A and B are arranged in series, A rejecting heat directly to B. Engine A receives 200kJ at a temperature of 421°C from a hot source, while engine B is in communication with a cold sink at a temperature of 4.4°C. If the work output of A is twice that of B find (a) the intermediate temperature between A and B, (b) the efficiency of each engine, and (c) the heat rejected to the cold sink.
Solution:
Example 2: A reversible heat engine operates between temperature T1 and T (T1>T). The energy rejected from this engine is received by s second reversible heat engine at the same temperature T. the second engine rejects energy at a temperature T2 (T2<T1). Show that (a) temperature T is the arithmetic mean of temperature T1 and T2 if the engine produce the same amount of work output, and (b) temperature T is the geometric mean of temperatures T1 and T2 if the engine produces the same cycle efficiency.
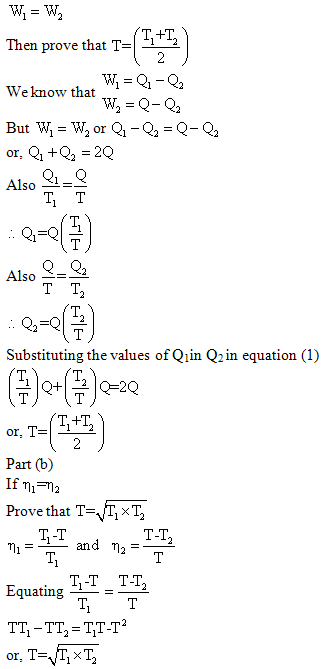 |
(1) (2) |
Example 3: A reversible engine works between three thermal
reservoirs, A, B and C. the engine absorbs an equal amount of heat from
the thermal reservoirs A and B kept at temperature TA and TB
respectively, and rejects heat to the thermal reservoir C kept at temperature
TC. The efficiency of the engine is times the efficiency of the
reversible engine, which works between the two reservoirs A and C. prove
that
Solution: