C-glycosylation or C-mannosylation: This is different from other types of glycosylation. In others there is bonding between carbon-nitrogen and carbon-oxygen but C-mannosylation is characterized by Carbon-carbon bonding. This process is carried out by enzyme C-mannosyltransferase (c-Mtf) which links C1 of mannose to C2 of indole ring of tryptophan. The enzyme identifies the precise sequence Trp-X-X-Trp and relocates a mannose residue from dolichol-P-Man to the first Trp in the sequence. C-mannosylation has been spotted in multiple cell lines (42) and rat liver microsomes.
Ubiquitinization: This is one another post-translational modification where ubiquitin is added to protein. Ubiquitin is the eukaryotic protein coded by 4 different genes in mammals UBA52,RPS27AUBB, and UBC. Protein is made of 76 amino acids and has a molecular mass of about 8.5 kDa. It is characterized by presence of C-terminal tail and 7 lysine residues. In ubiquitinization, basically, carboxylic acid of the terminal glycine from the di‐glycine motif in the activated ubiquitin forms an amide bond to the epsilon amine of the lysine in the modified protein. It marks the cellular protein for the process of degradation via proteosome, changes protein’s location, prevent or promote protein-protein interaction.
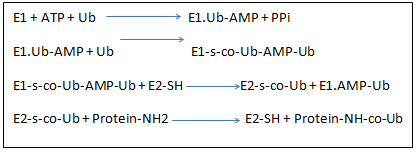
Steps followed in ubiquitinization process.
Activation of Ubiquitin: It occurs in a two-step reaction process. At first, ubiquitin interacts with ATP and forms ubiquitin-adenylate intermediate. In the next step, ubiquitin is transferred to E1 active site containing cysteine residue. This causes formation of thioester linkage between the C-terminal carboxyl group of ubiquitin and the E1 cysteine sulfhydryl group.
Transfer of Ubiquitin from E1 active site to E2 active site via trans-esterification reaction occurs.