O- Glycosylation: O-glycosylation is common for high molecular weight proteins found in mucus secretions and proteoglycan core protein that form extra-cellular matrix. O-glycosylation is also common in antibodies. Even though N-glycosylation has already occurred in these proteins it does not exclude O-glycosylation.
O-glycosylation occurs in Golgi apparatus after translation on serine and threonine side chains.O-glycosylation can also occur in the cytosol and nucleus to regulate gene expression or signal transduction through other Gtfs ,basic O gycosylation of serine or threonine are given below in Figure 31.5.
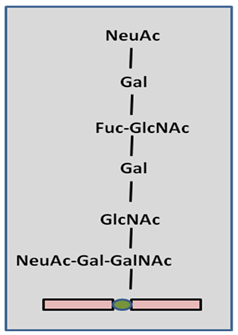
Figure 31.5: O-Glycosylation of threonine or serine
O-glycosylation is carried out by enzyme N-acetylgalactosamine (GalNAc) transferase, which transfers a single GalNAc residue to the β-OH group of serine or threonine. Some proteins are glycosylated with GalNAc, some with glucose, xylose, and mannose and so on. It is based on which cell type and species glycosylation is occurring. Sugar moieties are obtained from sugar nucleotides just like in N-glycosylation. O-glycosylation forms less complex structure compared to that in N-glycosylation.
Phosphoglycosylation: This type of modification is limited to parasites such as Leishmania andTrypanosomaand slime moulds like Dictyostelium. It is abundant in leishmania and is used to make proteophosphoglycans which is important for the organism to protect it from host’s defence mechanism. In this modification glycans are linked to serine and threonine residues by phosphodiester bond.