Glycan attachment: The pre-assembled glycan is attached to newly formed polypeptide chain via enzyme oligosaccharide transferase (OSTase). OSTase looks for consensus sequence Asn-X-Thr/Ser, where X may be any amino acid except proline. Once it is recognized then 14- mer precursor glycan is attached to carboxamido nitrogen on Asn of the developing polypeptide chain. β-OH group of serine and threonine residue acts as hydrogen bond donor for the reaction. Here in given figure we are discuss about the diagrammatical arrangement of glycogen arrangement. Glycan synthesis starts at the cytosolic face of the ER and when the structure is flipped into the ER lumen then it completed. Now OSTase enzyme transfer the precursor glycan to the Asn residue on the nascent protein as we discuss in given Figure 31.3.
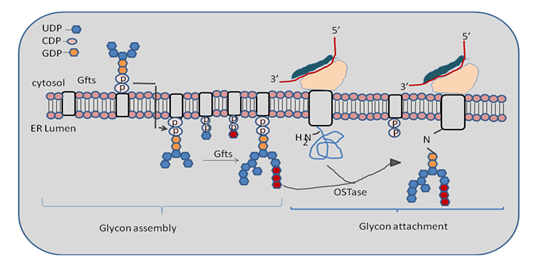
Figure 31.3: Glycon assembly and arrangement
Trimming in ER: Sugar molecules are trimmed off by glycosidases via hydrolysis. This process occurs both in golgi and ER and both serves different purposes. In ER, trimming is done to both screen protein folding and specify when proteins should be degraded. There is continuous cycle of removal and addition of terminal glucose molecules that enables proper protein folding. At first, two terminal Glc are removed followed by binding of calnexin and calreticulin to the chain via remaining Glucose molecules. These then act as chaperons for folding of proteins. Final Glc is hydrolysed by glycosidase II to discharge glycoprotein from chaperons. Folded output from this process has not yet acquired native conformation. Now, UDP-glucose glycoprotein glucosyltransferase acts on non-native folded protein and transfers a Glc to the glycoprotein, and the protein again is constrained to the lectin chaperones to aid proper protein folding. The cycle continues till the protein is properly folded. Properly folded proteins are trafficked to golgi bodies for further processing. Those proteins which are not properly folded are identified by ER-resident mannosidase (ERManI). These proteins are deglycosylated by glycanase N and then forwarded to ER-associated degradation (ERAD).