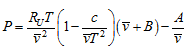Similarly, two different substances are considered to be in ‘corresponding states', if their pressure, volumes and temperatures are of the same fraction or multiples of the critical pressure, volume and temperature of those substances.
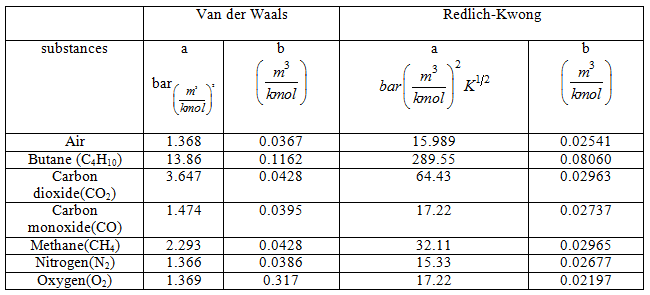
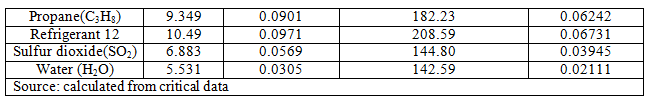
Values of constants 'a' and 'b' for van der Waals and Redlich-Kwong are given below (Table 2.2)
Table: 2.2 Values of a and b
The generalized compressibility chart in terms of reduced properties is shown in generalized compressibility chart. It is very useful in predicting the properties of substances for which more precise data are not available.
Other form of real gas equations in terms of reduced properties can be estimated similarly.
(2) Benedict-Webb-Rubin
The Beattie-Bridgeman equation of state is
|
(2.171) |
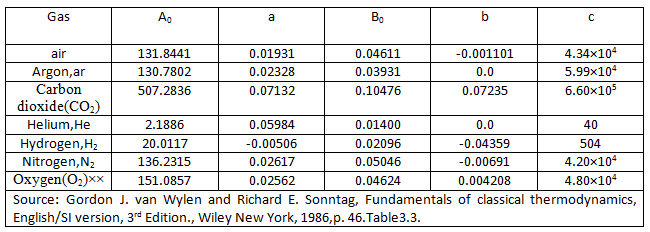
In this equation, P is in Kpa, ![]() is in m3/kmol, T is in K and Ru = 8.314Kpa.m3 / (kmol.K)
is in m3/kmol, T is in K and Ru = 8.314Kpa.m3 / (kmol.K)
where
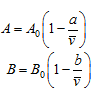
The five constants in the Beattie-Bridgeman equation are as follows (Table 2.3):
Table 2.3 Different Gas Constants