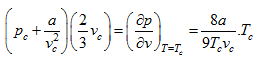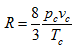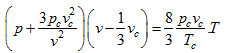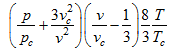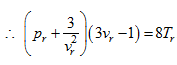From these two equation, by rearranging and dividing,
|
(2.163) |
Substituting the value of b in Eq. (2.65)
|
(2.164) |
Substituting the values of b and R in Eq. (2.152a)
|
(2.165) |
|
(2.166) |
Therefore, the value of R becomes
|
(2.167) |
The values of a, b, and R have thus been expressed in terms of critical properties, substituting these in van der Waals equation of state
|
(2.168) |
or,
|
(2.169) |
Using the reduced parameter
|
(2.170) |
In the reduced equation of state (2.170) the individual coefficients a, b and R for a particular gas have disappeared. So this equation is an expression of the law of corresponding states because it reduces the properties of all gases to one formula.