Comparing this equation and rearranging
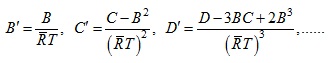
![]()
Therefore
Z |
(2.152) |
The terms ![]() etc. of the virial expansion arise on account of molecular interaction. If no such interactions exist (at very low pressures) B=0,C=0 etc. Z=1 and
etc. of the virial expansion arise on account of molecular interaction. If no such interactions exist (at very low pressures) B=0,C=0 etc. Z=1 and ![]() .
It may be observed from Eq (2.152) that the compressibility factor is a function of temperature and pressure of a gas.
.
It may be observed from Eq (2.152) that the compressibility factor is a function of temperature and pressure of a gas.
A graph for variation of pressure versus Z with constant temperature can be plotted from which value of Z for a pair of pressure and temperature states can be estimated. Further using the equation pv= ZRT, the volume of the gas can be obtained.
Example 1: Express the van der Waals equation of state in virial form.
Solution:
.jpg)
|
(2.152) |
The second virial coefficient ![]() the third virial coefficient c = b2, etc.
the third virial coefficient c = b2, etc.
From Eq. (2.59), on mass basis
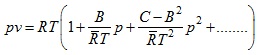
Example 2: Determine Boyle temperature from van der Waal's equation
Solution:
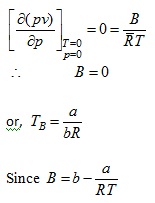
.jpg)