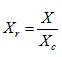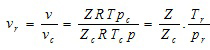The point at which B is equal to zero gives the Boyle temperature.
The second virial coefficient is the most important. Since 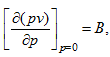 this B is known,
the behavior of the gas at moderate pressure is completely determined.
this B is known,
the behavior of the gas at moderate pressure is completely determined.
The terms which contain higher power ![]() becomes significant only at very high pressures .
becomes significant only at very high pressures .
LAW OF CORRESPONDING STATES
The general shapes of the vapour dome and the constant temperature lines on the p-v plane are similar for all substances. However, scales may be different for different substances. This similarity can be utilized with application of dimensionless properties called reduced properties.
A reduced property is the ratio of the existing property (X) to the critical property ( Xc ) of the substance. Thus
|
(2.153) |
Where the subscript r and c refer to the reduced and critical properties, respectively.
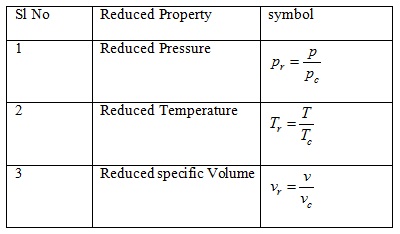
Various primary properties in terms of reduced property are given in Table 2.1 .
Table 2.1: Reduced Properties
The relations among the reduced properties, Pr , Tr , vr and vr is known as the law of corresponding states.
The specific volume or molal volume of different gases are different under same pressure and temperature. However, it is found from experimental data that at the same reduced pressure and reduced temperature, the reduced volumes of different gases are approximately the same. Therefore, for all substances
|
(2.154) |
Now,
|
(2.155) |
where ![]() is called the critical compressibility factor.
is called the critical compressibility factor.