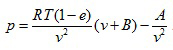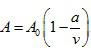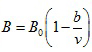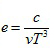Ideal gas equation of state is pv = RT.
This equation was established from the postulates of the kinetic theory of gases by Maxwell with the following assumptions:
(1) There is little or no attraction between the molecules of the gas
(2) Volume occupied by the molecules is negligibly small compared to the volume of gas
Applicability: Ideal gas equation of state is applicable to gas with very small pressure or very high temperature. With increase in pressure the intermolecular force of attraction and repulsion increases, and also the volume of the molecules becomes appreciable compared to the gas volume. Under such condition real gas equations are to be used.
Following are some of the real gas equations:
- van der Waals equation of
state for real gas
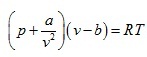
(2.138)
where
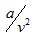 is accounting for the force of cohesion and b is accounting for volume of the molecules termed as co-volume .
is accounting for the force of cohesion and b is accounting for volume of the molecules termed as co-volume . - Berthelot equation of state
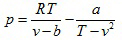
(2.139)
- Dieterici equation of state
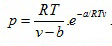
(2.140)
- Redlich-Kwong equation of state
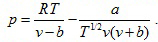
(2.141)
- Saha-Bose equation of the state
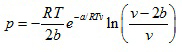
(2.142)
where a and b are constants which can be evaluated from the critical data.
-
The van der Walls, Berthelot and Dieterici equations of state are of limited accuracy.
-
The Redlich-Kwong equation gives good results at high pressures and is fairly accurate for temperatures above the critical value.
-
Saha-Bose equation of state is quite accurate for densities less than about 0.8 times the critical density
One more widely used equation of state with good accuracy is the Beattie-Bridgeman Equation:
|
(2.143) |
where
|
|
|
There are five constants, A0 , B0 , a, b, and c to be determined experimentally for each gas.