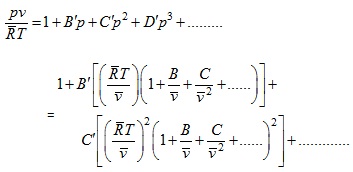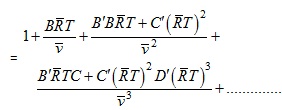Virial expression
The relation between pv and p in a term of power series may be expressed as
|
(2.144) |
For any gas
|
(2.145) |
|
(2.146) |
An alternative expression is
|
(2.147) |
Both expressions in Eqs.(2.146) and (2.147) are known as virial expansions or virial equations of state, first introduced by the Dutch physicist, Kammerlingh onnes B´, C´, B, C,…….. etc. are called virial coefficients. B´ and B are called second virial coefficients, C´ and C are called third virial coefficients, and so on. For a given gas, these coefficients are functions of temperature only.
The ratio ![]() is called the compressibility factor, Z. for an ideal gas Z=1. The magnitude of Z for a certain gas at a particular pressure and temperature gives an indication of the extent of deviation of the gas from the ideal gas behavior.
is called the compressibility factor, Z. for an ideal gas Z=1. The magnitude of Z for a certain gas at a particular pressure and temperature gives an indication of the extent of deviation of the gas from the ideal gas behavior.
The virial expansions become
|
(2.148) |
and
|
(2.149) |
The relations between B', C' and B, C… can be derived as follows
|
(2.150)
(2.151) |