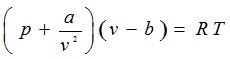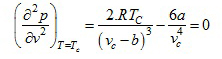From Eqs. (2.154) and (2.155),
|
(2.156) |
Experimental values of Zc for most substances fall within a narrow range of 0.20-0.30. Therefore, Zc may be taken to a constant.
Eq.(2.156) reduces to
Z = f (pr, Tr) |
(2.157) |
where Tr is plotted as a function of reduced pressure and Z, a single plot, known as the generalized compressibility chart, is found to be satisfactory for a great variety of substances. Although necessarily approximate, the plots are extremely useful in situations where detailed data on a particular gas are lacking but its critical properties are available.
Law of corresponding states for van der Waal's gas
For a van der Waals gas,
(2.158) |
where a, b, and R are the characteristic constants of the particular gas.
(2.159) |
(2.160) |
It is therefore a cubic profile in v and for given values of p and T has three roots of which only one need be real.
-
For low temperatures, three positive real roots exist for a certain range of pressure.
-
As the temperature increases the three real roots approach one another
-
At the critical temperature all the roots become equal. Above this temperature only one real root exists for all values of p.
-
the critical isotherm Tc at critical state on the p-v plane (fig.10.4),where the three real roots of the van der Waals Equation coincide, not only has a zero slope, but also its slope changes at the critical state (point of inflection), so that the first and second derivatives of p with respect to v at T = Tc are each equal to zero. Therefore,
(2.161) |
(2.162) |