Kinetics and mechanism
Ethylene oxide formation over silver catalysts is proposed to occur through dissociative chemisorption of oxygen. The proposed mechanism by Haul and Neubauer [4] is as follows:
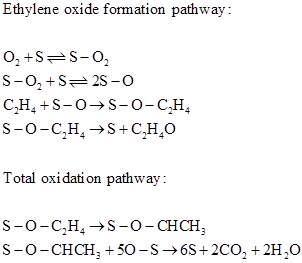
Park and Gau [5] proposed a Langmuir –Hinshelwood rate expression for supported silver over barium doped silica as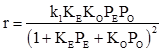
Here E = ethylene and O = oxygen
Deactivation
Unsupported silver catalysts are very sensitive to poisoning by S, N and halide compounds. Very high temperature also results in fusion of silver catalysts. Catalysts life for of Silver catalysts is only 6 months. Fe2O3-Co2O3-MoO3 is more rugged and last for about 1-2 years.
Book reference:
• C. H. Bartholomew and R. J. Farrauto, Fundamentals of Industrial catalytic Processes, Wiley, VCH, 2006
Journal reference :
1. J.A. Dumesic, D.F Rudd, L.M Aparicio, J.E. Rekoski and A.A. Trevino, The microkinetics of heterogeneous catalysis, ACS ,1993
2. R.D. Cortright, Per E. Levin, J. A. Dumesic, Ind. Eng. Chem. Res. 1998, 37, 1717-1723
3. P. Mars and J.G. Maessen, J. Catal. 10 (1968) 1257
4. R. Haul and G. Neubauer J. Catal. 105 (1987) 39
5. D.W. Park and G. Gau, J. Catal. 105(1987) 81